Introduction
Background
Sjögren syndrome is a slowly progressive inflammatory disorder that involves the exocrine glands.1 Mikulicz and others recognized the findings of keratoconjunctivitis and xerostomia as an entity in the late 1800s.2 In 1933, Sjögren recognized the association of this symptom complex with polyarthritis. Subsequent studies showed that Sjögren syndrome may be a primary disorder or may be secondary to other autoimmune disorders, such as systemic lupus erythematosus (SLE), rheumatoid arthritis, scleroderma, and biliary cirrhosis. Extraglandular manifestations may mimic these other autoimmune disorders.1 The primary disorder is most common in women in the fourth and fifth decades of life. Sjögren syndrome may overlap with other pediatric autoimmune disorders and, less commonly, may present as a primary condition.3
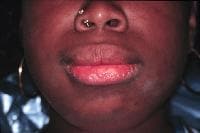
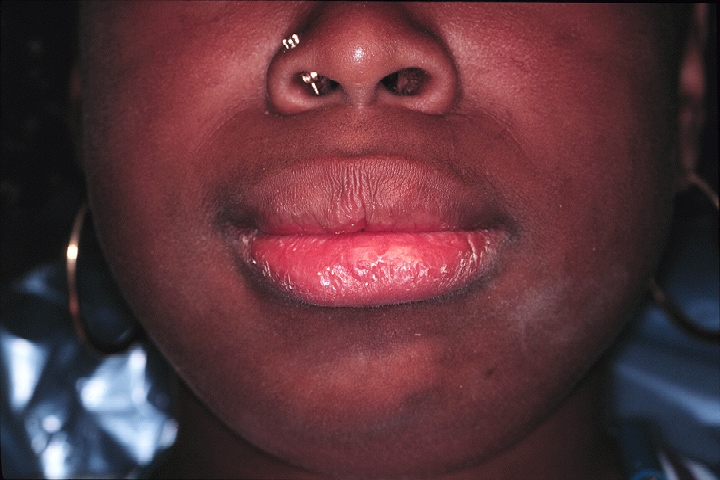
Lower facial appearance of a 14-year-old adolescent girl with Sjögren syndrome. She exhibits both parotid and submandibular gland enlargement and chapped lips.
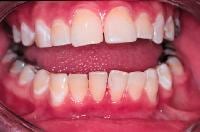
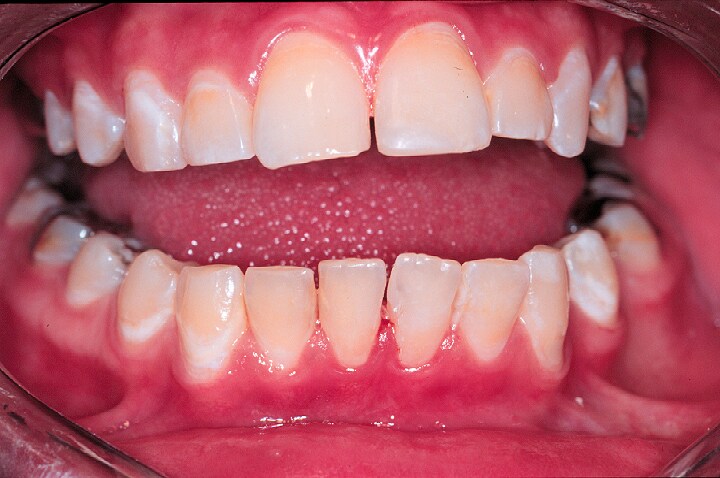
Intraoral view of a 14-year-old adolescent girl with Sjögren syndrome (see Media file 1). Hyposalivation results in erythema of the mucosa, gingivitis, decalcification or white spot lesions of the teeth at the cervical margin, and dental caries with extensive restorations of the posterior teeth.
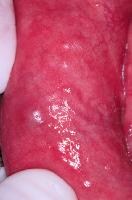
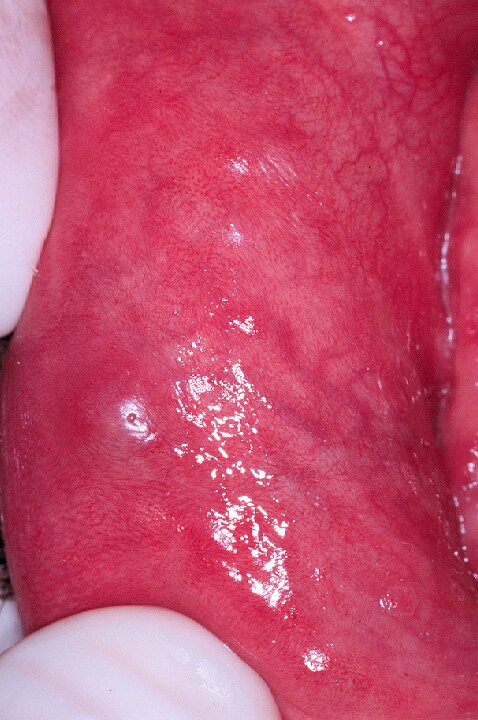
Erythema of the labial mucosa with enlargement of the minor salivary glands and superficial mucoceles.
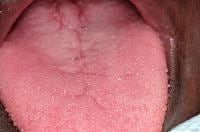
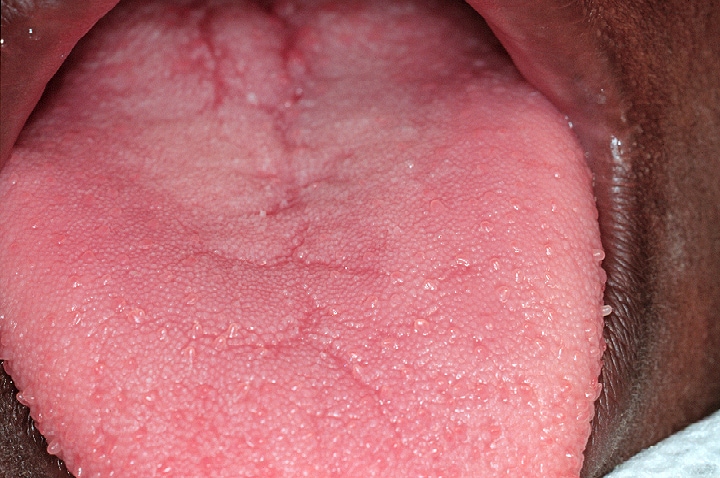
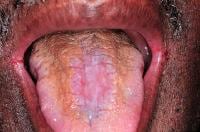
The dorsal surface of the tongue demonstrates generalized atrophy of the filiform papillae, mild fissuring, and median rhomboid glossitis.
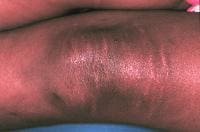
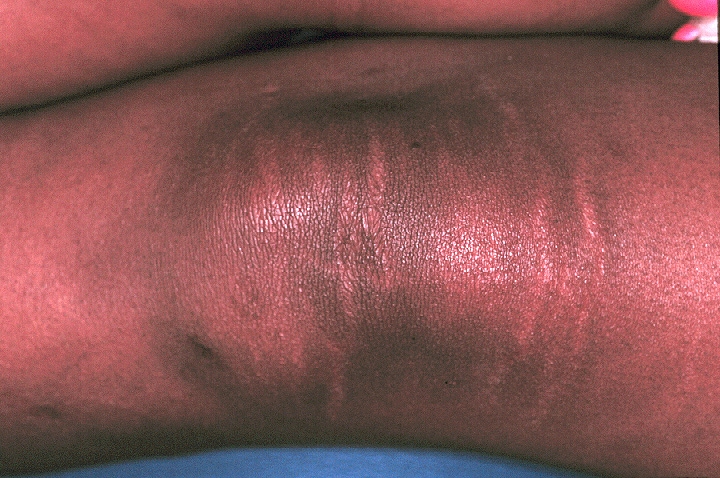
A 14-year-old adolescent girl with Sjögren syndrome (see Media file 1) with painful unilateral swelling of the knee and hyperpigmentation of the overlying skin.
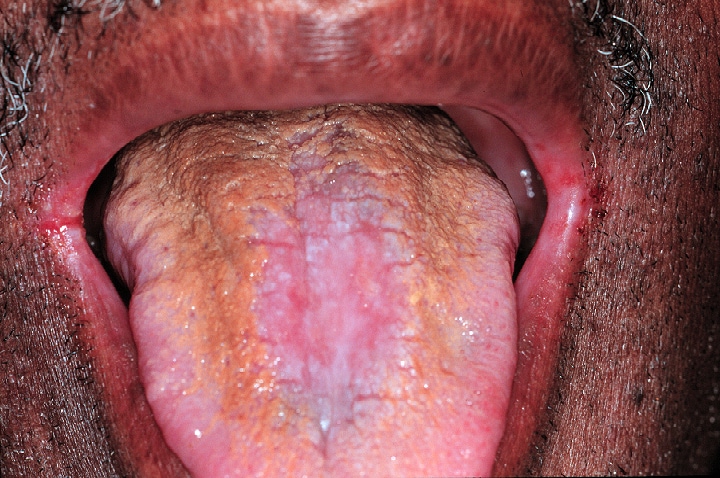
The dorsal tongue demonstrates hyperplastic candidiasis with focal erosions and a brown hairy tongue. Ulcerated fissures are observed on the corners of the mouth that represent angular cheilitis.
Pathophysiology
Lymphocytic infiltration in the glandular lobules is the characteristic abnormality in lacrimal or salivary gland biopsy findings. The disorder appears to be mediated by a complex cascade of immune events and is often characterized as an autoimmune exocrinopathy.4,5
The predominant immune cells are activated memory CD4+ T cells that express alpha/beta T-cell receptors, suggesting a central role in the pathogenesis of this disorder.4 Activated B cells are also found in the lesions, which may be responsible for the production of the autoantibodies to autoantigens Ro (SS-A) and La (SS-B). Anti-Ro (SS-A) is found in 40-45% of adult patients with Sjögren syndrome, and anti-La (SS-B) is found in 50% of these patients. Positivity in these antibodies appears to be higher in children, according to a systematic review of the literature (rheumatoid factor [RF], 66%; antinuclear antibody [ANA], 78%; SS-A, 74%; SS-B, 65%).6 Because these autoantibodies are also observed in other autoimmune disorders, including SLE, further diagnostic tools must be used for a more definitive diagnosis.
Family members of these patients have a higher incidence of Sjögren syndrome. The association of Sjögren syndrome with human leukocyte antigen (HLA)–B8, HLA-Dw3, HLA-DR3, and with the DQA1*0501 allele supports the notion of genetic susceptibility.7
An active area of research involves genetic polymorphisms of cytokine genes, including IL10 and TNFA.4 Associations with these polymorphisms might implicate inappropriate regulation of the immune response in Sjögren syndrome. Altered apoptotic mechanisms have also been implicated in the pathogenesis of Sjögren syndrome–related exocrinopathies. Exocrine gland tissue damage and chronic inflammation leads to fibrotic changes and impaired glandular function. The pathophysiology of extraglandular manifestations is thought to be due to similar immune mechanisms.
Frequency
United States
Although Sjögren syndrome has been recognized in patients of all ages, it primarily affects women in the fourth and fifth decades of life. Overall, Sjögren syndrome is the second most common autoimmune disorder in adults, after rheumatoid arthritis. In the United States, between 500,000 and 2 million adults have this disease.2
It is rare in childhood, although over 145 cases of primary disease have been described in the international pediatric literature.6 Five percent of adult patients report onset of symptoms before age 12 years. Clinical manifestations and classification schema differ in pediatric patients; therefore, this disorder may be underreported. Features of Sjögren syndrome are well described in pediatric patients who have other autoimmune disorders.
International
Epidemiological studies from different ethnic groups show prevalence rates similar to those in the United States.
Mortality/Morbidity
Although most adult patients have a mild and benign course, they often experience painful eye irritation, severe dental caries, and dyspareunia. Pediatric patients often do not have early sicca syndrome symptomatology but often have recurrent parotitis. Because of the insidious nature of these symptoms, patients often do not seek medical attention until more severe symptoms appear years later. The time from disease onset until diagnosis is often 9 years in adults and perhaps as long as 3 years in pediatric patients. In a multicenter retrospective review of 40 patients, few major complications were reported from time of onset (follow-up, 0-7.5 y).8 According to another small report of pediatric patients with at least 3 years follow-up, minimal progression in disease status was reported.9 Some pediatric patients may develop systemic features that involve the nervous system, kidney, and lungs.10,8,11
Lymphoproliferative disorders increase 40-fold in adult Sjögren syndrome. Although significant lymphoproliferation usually remains confined to the salivary and lacrimal glands, extraglandular lymphoproliferation (eg, lymphadenopathy, hepatosplenomegaly) sometimes resembles lymphoma (eg, pseudolymphoma) but may herald frank malignancies, including non-Hodgkin lymphoma, Waldenström macroglobulinemia, and B-cell lymphoma. Associations with lymphoproliferative diseases remain unclear in pediatric populations.
Sex
The female-to-male ratio is approximately 9:1 in adults.12 This female-to-male ratio was 7:1 in one multicenter international pediatric cohort and 6:1 in a systematic review of the literature.8,6
Age
Sjögren syndrome is relatively rare in childhood. The mean age of diagnosis in primary juvenile Sjögren syndrome is 10 years in 2 studies.6,8 The peak incidence in adult populations is around the fourth and fifth decades of life.
Clinical
History
Clinical manifestations of pediatric Sjögren syndrome may vary more than those seen in adults with the disease.9 The constellation of symptoms seen in children (eg, lower frequency of sicca syndrome, higher rates of parotid enlargement, higher prevalence of immunologic markers) may be similar to those found in young adult patients (ie, <35>13
- Sicca syndrome
- Keratoconjunctivitis: Symptoms include dry eyes with reduced tear production, gritty or sandy sensation under the lids, red eyes, and photosensitivity. Keratoconjunctivitis is less prominent in primary juvenile Sjögren syndrome.6,8,14 Lacrimal gland enlargement appears to be a feature in both primary and secondary pediatric Sjögren syndrome.
- Xerostomia: Symptoms include decreased saliva production that leads to difficulties with chewing, swallowing, and even speech; abnormality in taste and smell; dental caries; mucosal burning sensation; sensitivity to spicy and acidic foods and beverages; increased risk for oral candidiasis; hoarseness of voice, and dysphonia (common in adults). Recurrent parotitis appears to be the most common oroglandular manifestation in pediatric populations.6,8
- Musculoskeletal symptoms
- Arthralgia (often noninflammatory), morning stiffness, nonerosive arthritis
- Myalgia, muscle weakness
- Cutaneous findings
- Raynaud phenomenon
- Nonthrombocytopenic purpura, especially of lower extremities
- Nasal, vaginal, and cutaneous dryness
- GI symptoms
- Dysphagia, nausea, epigastric, and abdominal pain
- Achalasia (in children)
- Achlorhydria, chronic atrophic gastritis (adult patients)
- Primary biliary cirrhosis
- Pulmonary findings
- Dyspnea due to mild interstitial disease
- Dry cough
- Renal findings - Interstitial nephritis and renal tubular acidosis
- Other symptoms - Fatigue, depression, and insomnia
Physical
- Parotid gland enlargement and recurrent parotitis (most prominent feature in pediatric populations)6,8,14
- Corneal ulceration, vascularization, uveitis
- Vasculitic lesions - Purpura and erythema nodosum
- Lymphadenopathy
- Autoimmune thyroiditis
- Nervous system manifestations
- Peripheral sensorimotor neuropathy
- CNS disorders such as movement disorder, transverse myelopathy, encephalopathy, aseptic meningitis, dementia, optic neuropathy, and cranial neuropathies (in both adult and pediatric patients)
- Musculoskeletal manifestations
- Intermittent synovitis
- Chronic nonerosive oligoarticular or polyarthritis (Jaccoud arthropathy is observed in adults.)
- Myalgias
- Oral cavity manifestations
- Mild erythema and thinning of the mucosa
- Dental caries
- Traumatic erosions and ulcers, angular cheilitis, and chapped lips
- Frothy, ropey, and thickened saliva
- Erythema, fissuring, coating, and depapillation of the dorsal tongue
- Halitosis
Causes
Multiple etiological factors are likely involved in the pathogenesis of Sjögren syndrome.4,1
- Sex hormone: As with other autoimmune disorders, Sjögren syndrome is more common in females. This suggests that sex hormones may play a role in shaping the autoimmune response.
- Genetics: The association of certain human leukocyte antigens (HLAs) suggests that genetic factors likely play a role in the pathogenesis of Sjögren syndrome. Patients with HLA-B8, HLA-Dw3, and HLA-DR3 have a high incidence of autoantibody production and extraglandular manifestations. Most patients also have the DQA1*0501 allele, which may play a role in the pathogenesis of this disease.
- Environmental factors: Epstein-Barr virus (EBV) replicates in the salivary glands during primary infection and remains latent in these organs. EBV-DNA is recovered from salivary glands and saliva of these patients. Its etiopathological role cannot be proven. Human immunodeficiency virus (HIV), human T-cell leukemia-lymphoma virus type 1 (HTLV-1), and cytomegalovirus (CMV) are also under scrutiny as possible inciting agents in adult patients that may lead to lymphoproliferation observed in the end organs.
- Autoantibodies: The presence of anti-Ro (SS-A) and anti-La (SS-B) is associated with earlier onset, longer duration, and more extensive extraglandular manifestations of primary Sjögren syndrome. Antinuclear antibody (ANA), rheumatoid factor (RF), anti-Ro (SS-A) and anti-La (SS-B) are prevalent in pediatric cohorts.6,12,8 Researchers have found a high prevalence of anti-Muscarinic-3 acetylcholine receptor autoantibodies in Japanese children with primary Sjögren syndrome.15 These antibodies are postulated to be involved in defective gland function.
- Autoantibody production and evidence of a polyclonal gammopathy implicate B-cell dysfunction as a component of Sjögren syndrome pathogenesis. Increased levels of B-cell activating factor (BAFF) or B-lymphocyte stimulator (BLyS), which are essential for B-cell survival, can be found in patients with Sjögren syndrome. Thus, B-cell depletion therapies that include the anti-CD20 monoclonal antibody (rituximab) are being used in open-labelstudies.16
- Inflammatory reactivity: In situ evidence suggests that proinflammatory cytokines, interleukin (IL)–1, IL-6, and tumor necrosis factor (TNF)–alpha are produced in the salivary glands. These cytokines are found in the infiltrating lymphocytes and the epithelial cells. This suggests that the infiltrating lymphocytes (ie, predominantly CD4 T cells) play a role in the perpetuation of an aberrant immune response. Specific IL-10 haplotypes are associated with early onset and vasculitic features of disease. Additional cytokine and chemokine gene polymorphisms are currently being studied.
Herbal Treatment for Sjogren’s Syndromeinformation about the Symptoms, Causes and Diagnosis. Natural Herbal Treatment for Sjogren’s Syndrome with Herbal Product Neteton Natural Supplement for dryness of the mouth and eyes.
Trả lờiXóaI was diagnose with genital warts since 2012 i have be taking lot treatment and all i got is outbreak. in 2015 I gave up the treatment because I can't continues wasting time and money on treatment at the end it will not cure me. about 6 weeks ago i did natural research online I had So many people talking good about natural remedy, after the research i was recommended to Dr onokun, And I wrote to him through his email and told him my problem after some conversations with him he gave me natural treatment after 1 week Dr onokun treated me i got cured permanently. and i went to see my doc he confirmed that the diseases has gone out from my body. every patients should know there is 100% natural hpv cure. contact Dr onokun his email address: dronokunherbalcure@gmail.com
Trả lờiXóa